By Dr. Jerry Shurson, University of Minnesota Department of Animal Science © 2019 Feedstuffs. Reprinted with permission from Vol. 91, No. 07, July 1, 2019
Regulations that have led to restricting antibiotic use for growth promotion in livestock have caused swine nutritionists to consider various feed additives, functional ingredients and functional nutrients that can be used as alternatives to promote pig growth and health.Compared with other grain byproducts, corn dried distillers grains with solubles (DDGS) have relatively high concentrations of natural antioxidant compounds that may contribute to reducing oxidative stress. Furthermore, DDGS contains about 10% spent yeast, and yeast cell walls contain beta-glucans, mannanoligosaccharides (MOS) and nucleotides that have been shown to provide beneficial effects for swine health and growth performance.
The relatively high concentration of insoluble fiber in DDGS may affect gut health through changes in cell proliferation, digesta viscosity and the microbiome, but limited studies have been conducted to characterize these effects. Although the direct effects of antioxidants, yeast cell wall components and fiber on swine health are not well understood, a few studies have shown gut health benefits for specific pathogens when feeding DDGS diets to pigs.
Finally, a new dimension of sourcing feed ingredients involves assessing the relative risk of transmission of foreign animal diseases such as porcine epidemic diarrhea virus (PEDV) and African swine fever virus (ASFV). Preliminary data suggest that, compared to soybean meal and corn, DDGS is a lower-risk factor for PEDV survival and does not appear to be a significant risk factor for ASFV transmission.
Phytochemicals, antioxidants
Several studies have been conducted to determine the concentrations of phyto-chemicals and antioxidant capacity in DDGS.Winkler et al. (2007) used several different extraction methods to quantify the phytosterols (1.97-2.91 mg/g of DDGS), phytostanols, total tocopherols and tocotrienols (0.73-1.82 mg/g of ex- tract) and ferulate phytosterol esters (0.35-0.53 mg/g of DDGS) in corn oil and corn fiber oil from DDGS.
In a subsequent study, Winkler-Moser and Breyer (2011) determined the oxidative stability, tocopherols, tocotrienols, carotenoids, phytosterols and steryl ferulates in corn germ oil and oil extracted from post-fermentation fractions and DDGS. They found that hexane-extracted oil from DDGS was the most oxidatively stable and had significant quantities of functional lipids that serve as antioxidants to increase the oxidative stability. Luthria et al. (2012) also determined the phenolic (vanillic, caffeic, p-coumaric, ferulic and sinapic) acid concentration and antioxidant capacity of DDGS and compared those to the concentrations in corn grain. They found that the phenolic acid profile in DDGS was similar to corn, with ferulic and p-coumeric acids representing about 80% of the total phenolic acids. Furthermore, the total phenolic acid content in DDGS was 3.4- fold greater, and the antioxidant capacity was 2.6-fold greater than in corn grain.
In a subsequent study, Luthria et al. (2014) reported that the increased phenolic acid concentration in DDGS was mainly due to depletion of starch during the ethanol fermentation process.
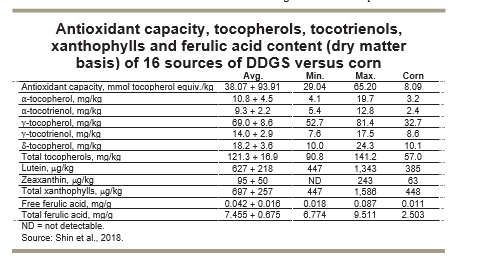
More recently, Shin et al. (2018) evaluated the antioxidant capacity, tocopherols, tocotrienols, xanthophylls and ferulic acid content of 16 sources of DDGS and compared those values with corn grain (Table). The antioxidant capacity of DDGS sources ranged from 29 to 65 mmol tocopherol equivalent per kilogram. Total tocopherols ranged from 91 to 141 mg/kg, total xanthophylls ranged from 447 to 1,586 µg/kg and total ferulic acid content ranged from 6.8 to 9.5 mg/kg.
As expected, DDGS sources had much greater concentrations of these important functional lipids than corn grain. Tocopherols, carotenoids (xanthophylls) and ferulic acid are strong antioxidant compounds that appear to be beneficial in preventing greater oxidation during thermal exposure, which occurs during the DDGS production and drying processes. Furthermore, the relatively high concentrations of these compounds in corn oil from DDGS appear to be beneficial in minimizing oxidative stress when feeding highly oxidized DDGS sources to nursery pigs (Song et al., 2013; Hanson et al., 2015b) and wean/finish pigs (Song et al., 2014).
It is important to determine the extent of lipid oxidation and antioxidant capacity in feed ingredients, especially those with relatively high lipid concentrations (e.g., DDGS), because it can affect the potential for further oxidation, storage stability and the need for adding supplemental antioxidants. Hanson et al. (2015a) showed that adding either ethyoxyquin or TBHQ antioxidants were about 50% effective at preventing further oxidation from occurring when DDGS samples were stored under hot, humid conditions.
Recent reviews have been published regarding the complexity of measuring and interpreting lipid oxidation data (Kerr et al., 2015; Shurson et al., 2015). However, a meta-analysis of several studies has shown significant reductions in growth performance and metabolic oxidation status in pigs fed oxidized lipids (Hung et al., 2017).
Although results from previous studies (Winkler et al., 2007; Winkler-Moser and Breyer, 2011; Luthria et al., 2012; Shin et al., 2018) consistently showed that DDGS has a high antioxidant capacity, Song and Shurson (2013) evaluated the extent of corn oil oxidation in 31 corn DDGS sources. The results showed that oil oxidation occurs during the DDGS production and drying process, and the corn oil in some sources may be up to 25 times more oxidized than the oil in corn grain. In a subsequent study, Song et al. (2013) fed 0% or 30% DDGS diets containing three dietary levels of supplemental vitamin E — none, a level at the National Research Council [NRC} requirement and a level 10 times the requirement — to evaluate the oxidative status of weaned pigs. The DDGS used in this study was the most oxidized source identified in the Song and Shurson (2013) study, which also contained 0.95% sulfur. Results showed that feeding the DDGS diet increased serum alpha-tocopherol and sulfur-containing amino acid concentrations, liver glutathione concentration and glutathione peroxidase activity. They concluded that the increased concentrations of sulfur-containing antioxidants (methionine, taurine and glutathione) may protect pigs against oxidative stress when fed highly oxidized DDGS and that increasing dietary vitamin E concentrations may not be necessary.
Several years ago, a few veterinarians observed an increased incidence of vitamin E and selenium deficiency known as mulberry heart disease (MHD) in nursery pigs and attributed this to feeding DDGS diets. Therefore, Hanson et al. (2015b) conducted a study to determine if feeding diets containing the most oxidized DDGS source (identified by Song and Shurson, 2013), with or without supplemental vitamin E (five times the requirement), to gestating and lactating sows and their offspring through the nursery period caused an increase in MHD during the nursery period.
Pigs fed the 30% oxidized DDGS diet had increased feed intake, but average daily gain was not affected compared to those fed the corn/soybean meal control diet. Furthermore, feeding the oxidized DDGS diet, with or without high vitamin E supplementation, increased serum alpha-tocopherol concentrations, but there were no differences in glutathione peroxidase activity and TBARS concentrations, nor were there any histopathological lesions indicative of MHD when pigs’ hearts were examined.
Similar to the results reported by Song et al. (2013), the serum concentrations of total sulfur-containing amino acids (cystathione, cysteine, methionine and taurine) were increased 40-50% in pigs fed the oxidized DDGS diets. Sulfur-containing amino acids have metabolic antioxidant properties and can counteract oxidative stress by acting as reducing agents.
Results of this study suggest that the increased intake of sulfur-containing amino acids, vitamin E and other antioxidant compounds in DDGS prevented the development of MHD and the potential negative effects of feeding oxidized DDGS.
Weber and Kerr (2011) evaluated the effects of feeding DDGS diets on oxidative stress and immune function in growing pigs. They found no effects on circulating metabolic oxidative stress indicators from feeding a 35% DDGS diet but showed an increase in plasma immunoglobulins A and G, suggesting an improvement in humoral immunity in finishing pigs.
In contrast, Li et al. (2012) reported that feeding DDGS diets to growing/finishing pigs reduced plasma and tissue redox status. However, Song et al. (2014) showed no growth performance benefit from adding supplemental vitamin E to highly oxidized DDGS diets fed to wean/ finishing pigs.
Therefore, the majority of studies indicate that feeding diets containing DDGS with varying amounts of oxidized lipids has no detrimental effect on growth performance and may enhance humoral immunity and minimize oxidative stress due to increased antioxidant compounds in DDGS.
Yeast content in DDGS
Significant amounts of yeast are added to fermenters during the ethanol production process, and spent yeast remains in co-product streams used to produce DDGS. Yeast cell walls contain beta-glucans, MOS and nucleotides, which are well-known nutraceutical compounds that have generally been shown to improve animal growth performance and health (Shurson, 2018).However, it is difficult to quantify the concentration of spent yeast and yeast cell wall components in DDGS (Shurson, 2018). A few researchers have attempted to estimate the yeast content in DDGS, but the methods used have several limitations, and the results reported have been highly variable.
Ingledew (1999) used a mass balance approach to estimate that 3.9% of the total DDGS biomass and 5.3% of the total protein content were derived from yeast. Belyea et al. (2004) estimated the yeast content of DDGS by calculating the proportional contributions of amino acids from yeast and corn relative to the total amino acid content in DDGS. They estimated that about 50% of the protein in DDGS was derived from yeast. However, this method ignored the effect of amino acid composition in corn, which resulted in an inaccurate estimate of the yeast content in DDGS.
Han and Liu (2010) used a more detailed approach by conducting a multiple linear regression analysis using the relative percentage of individual amino acids in corn and yeast relative to the total amino acid content of DDGS. They suggested that about 20% of DDGS protein is derived from yeast but acknowledged that this estimate may not be accurate because it assumes that 20% of DDGS biomass is comprised of yeast, which is not accurate.
Alternatively, it is possible to use the mannan content in yeast cell walls to calculate an estimate of yeast content in DDGS because mannans are only found in yeast cell walls and not in corn grain (Shurson, 2018). Using this method, the estimated yeast content of DDGS is about 10%, and the yeast content of high- protein corn co-products may be as high as 29%, depending on the production method used.
Yeast cell walls comprise about 15- 20% of the dry weight of yeast cells, and the main polysaccharide fractions are beta-glucans and mannans. The beta-glucans have been shown to adsorb or bind toxins, viruses and pathogenic bacteria (Vetvicka et al., 2014). Immune cells (macrophages) have receptors for beta-1,3/1,6-branched glucans, and their mode of action has been well defined in human nutrition and medicine (Rop et al., 2009). There is also research evidence showing that dietary beta-glucans may improve immune competence in young animals (Saeed et al., 2014).
Lim et al. (2009) used an enzymatic method designed to measure the beta-glucan content in barley and oats and estimated that the beta-glucan content of DDGS is about 0.57%. However, it is uncertain if this method is suitable for estimating the beta-glucan concentration in DDGS.
Kim et al. (2008) determined the total glucan concentration in DDGS to be about 21.2% (dry matter basis), 16% of which was attributed to cellulose and 5.2% from starch. Glucans are present in bacteria, fungi, yeast and cereal grains but vary in molecular configuration, solubility and functionality. Furthermore, cellulose is not considered a functional source of beta-glucans because it is in- soluble and does not provide the same functionality of yeast beta-glucans (Siko- ra et al., 2013).
No other studies have been conducted to estimate the beta-glucan content in DDGS, but recent estimates using a Mega-zyme International Yeast beta-glucan assay showed that the beta-glucan content in a high-protein DDGS was about 8.3% (Shurson, 2018).
MOS functions as a prebiotic — sources of nutrients for certain microbes in the gastrointestinal tract — that can subsequently result in having a probiotic effect, according to Spring et al. (2015), who reviewed results from numerous published studies that evaluated feeding MOS-supplemented diets to multiple animal species, and results generally showed improvements in growth rate, feed conversion and reductions in mortality. The researchers suggested that these improvements are a result of MOS binding and limiting the colonization of pathogens in the gastrointestinal tract, improving the integrity of the intestinal mucosa and modulating immune system activity and may be involved in antioxidant and anti-mutagenic defenses.
Alizadeh et al. (2016) used a modified procedure to determine the mannose content of a wheat/corn DDGS source and reported that it contained 1.6% man-nose.
Yeast contains significant amounts of nucleotides, with concentrations of total nucleic acids reported to be between 3.3% and 9.5% (Bacha et al., 2013) and 7% and 12% (Halasz and Lasztity, 1991). Alizadeh et al. (2016) determined the concentration of nucleotides to be 0.13% in a wheat/corn DDGS source, but no studies have been conducted to determine the nucleotide content in corn DDGS.
There is increasing research evidence that supplementing monogastric diets with nucleotides may affect the animals’ intestinal morphology and function, immune response, intestinal microbiota composition, liver function and morphology as well as growth performance (Sauer et al., 2011).
Microbial population changes
There is much interest in determining and understanding shifts in the gut microbiome of pigs that occur based on diet composition and interventions in swine health and growth performance. However, the current understanding of positive and negative effects of changes in microbial populations in the gastrointestinal tract on metabolism, nutrient utilization and the immune system is unclear.Tran et al. (2012) fed weaned pigs diets containing up to 30% DDGS and evaluated patterns of change in fecal microbial populations over time using an electrophoretic fingerprinting technique. Results showed that the 30% DDGS diet resulted in a more homogeneous microbial population with fewer bacterial species, but there were no changes in serum immunoglobulin concentrations. The researchers suggested that less microbial diversity in the gut microbiome may be associated with increased microbial eco-system instability, but there is no direct evidence to support this.
Ewing and Cole (1994) suggested that a greater ratio of Lactobacillus to Enterobacteriaceae spp. could be considered as an index of more favorable gut health conditions because Lactobacillus functions as a major component for preventing infections, while Enterobacteriaceae (including Escherichia coli) are detrimental to gut health.
Yang et al. (2010) determined the effects of feeding corn DDGS, wheat DDGS or a corn/wheat DDGS source on bacterial profiles in digesta collected at the terminal ileum of pigs. Their results showed that feeding the corn DDGS diet increased Enterobacteriaceae and tended to increase the number of Lactobacillus compared with feeding the wheat and corn/wheat DDGS sources. However, the Lactobacillus-to-Enterobacteriaceae ratio was greater in digesta of pigs fed the wheat DDGS compared with corn DDGS, but the lactic acid concentration was greater from feeding corn DDGS versus wheat DDGS. The researchers suggested that an increase in lactic acid production may potentially inhibit pathogen growth but provided no direct evidence to support this.
Intestinal cell differentiation
The small intestine contains many types of cells that have different functions. Enterocytes are involved in nutrient absorption, while other cells (goblet, endocrine, Paneth, tuft and M-cells) have secretory functions. Therefore, understanding the role of different dietary fiber sources (e.g., DDGS) on changes in intestinal cell composition, nutrient transporters and receptors, cell differentiation and the immune system is necessary to improve the nutritional efficiency and gut health of pigs.Saqui-Salces et al. (2017) fed finishing pigs a corn/soybean meal diet or diets containing similar neutral detergent fiber contents (about 21%) provided by wheat straw, DDGS or soybean hulls for 14 days. Their results showed that feeding diets containing DDGS and wheat straw had a greater effect on modulating intestinal cell differentiation by promoting goblet cells and altering expression of nutrient receptors and transporters than the diets containing soybean hulls or corn/soybean meal.
In a subsequent study, Vila et al. (2018) showed that feeding diets containing DDGS or wheat middlings increased the expression of MUCIN 2 (mucin) in the ileum without affecting the proportion of goblet cells compared to feeding a corn/ soybean meal diet. They also evaluated ileal gene expression of 12 cytokines to determine if feeding these high-fiber diets, with or without non-starch polysaccharide-degrading enzymes, changed the pro-inflammatory and anti-inflammatory responses. There were no effects of feeding DDGS or wheat midds diets on expression of the cytokines interferon- gamma, tumor necrosis factor-alpha, interleukin (IL) 1 beta, IL-2, IL-6, IL-8, IL-10, IL-12p40 and IL-23.
These responses were different from those reported by Weber et al. (2008), who showed that feeding nursery pigs a 7.5% DDGS diet for seven days increased ileal gene expression of IL-6, IL-1 beta and IL-10. However, the addition of enzymes to DDGS and wheat midds diets did affect the local immune profile of the ileum and favored a pro-inflammatory response. More efforts are needed to study the many complex responses from feeding DDGS diets on functional changes in gastrointestinal tract relative to nutrient utilization and the immune system.
Pathogen challenges
Lawsonia intracellularis. There is some evidence that feeding DDGS to swine has beneficial effects on the gut health of pigs infected with Lawsonia intracellularis.Whitney et al. (2006a,b) conducted two studies to evaluate the effects of feeding DDGS diets to growing pigs under a moderate L. intracellularis disease challenge. In the first study (2006a), pigs were fed diets containing 0% or 10% DDGS with and without antimicrobials (bacitracin methylene disalicylate and pulse dosing of chlortetracycline) for four weeks before infection and 21 days after infection. Feeding the DDGS diet decreased the prevalence, length and severity of lesions in the ileum and colon, and pigs fed the antimicrobial also had reduced prevalence and severity of lesions. Although the length, severity and prevalence of lesions were not affected in challenged pigs fed the DDGS diet with antimicrobials, fecal shedding of L. intracellularis was decreased by 14 days postchallenge.
These results suggest that DDGS may provide similar benefits to the antimicrobial used in this study for growing pigs challenged with this common pathogen.
However, in a subsequent study, Whitney et al. (2006b) evaluated the effects of feeding diets containing 20% DDGS, 5% soybean hulls or soybean hulls sprayed with a polyclonal antibody product to pigs challenged with L. intracellularis. In this study, lesion length, severity, prevalence and fecal shedding were generally not affected by diet, but lesion length and severity tended to be reduced in pigs fed the DDGS diet versus the diet containing the L. intracellularis polyclonal antibody product. The DDGS diet had no effect on serum immunoglobulin concentrations compared with challenged pigs fed the other dietary treatments.
The mechanisms of these responses are unknown because there is limited information on how feeding DDGS may affect the intestinal microbiota of pigs and their susceptibility to infection or colonization with pathogens.
Salmonella. Rostagno et al. (2013) conducted two experiments to determine if diets containing 20%, 30% or 40% DDGS affected susceptibility to, intestinal levels of and shedding of salmonella. In one experiment, pigs infected with salmonella and fed the control diet without DDGS had higher salmonella shedding frequency than pigs fed the 30% DDGS diet, but the overall responses suggest that diets containing DDGS do not alter the susceptibility to salmonella colonization in growing/finishing pigs.
Brachyspria. Brachyspira spp. are comprised of a diverse group of Gram-negative spirochetes, with Brachyspira hyodysenteriae known to cause swine dysentery in growing/finishing pigs. In addition, Brachyspira hampsonii can cause disease similar to B. hyodysenteriae, with increased mucosal thickening, hemorrhage and large amounts of mucus in the large intestine.
Wilberts et al. (2014) hypothesized that the high insoluble fiber content in DDGS may enhance the intestinal environment to encourage infection from pathogenic Brachyspira spp. and cause clinical colitis. To test this hypothesis, four-week-old pigs were inoculated with an uninfected dose or inoculum containing Brachyspira intermedia, Brachyspira pilosicoli, B. hampsonii or B. hyodysenteriae and were fed diets containing 0% or 30% DDGS for two weeks prior to inoculation through 21 days post-inoculation.
Results from this study showed that although feeding DDGS may increase the incidence of swine dysentery, it may not affect the severity. It appears that feeding a 30% DDGS diet may increase the risk of pigs developing swine dysentery, and when exposed to these pathogens, they may shed the pathogen one day earlier and develop dysentery almost twice as fast as those fed the 0% DDGS diet.
Risk of virus transmission
Foreign animal diseases, such as foot and mouth disease and classical swine fever, have had devastating impacts on global food animal production, trade and economics (Yang et al., 1999; Huang et al., 2000; Stegeman et al., 2000; Thompson et al., 2002; Waage et al., 2008).The introduction of PEDV into the U.S. pork industry in 2013 resulted in a loss of 7 million pigs, representing 10% of annual production (Schulz and Tonsor, 2015). Although the specific cause of PEDV introduction has not been conclusively determined, contaminated feed and feed ingredients can serve as sources of transmission of PEDV (Dee et al., 2014) as well as other coronaviruses (Trudeau et al., 2017), such as transmissible gastroenteritis virus (TGEV) or porcine del- ta coronavirus (PDCoV).
Dee et al. (2015) showed that PEDV was detected by virus isolation or a bioassay up to 30 days post-inoculation from soybean meal, DDGS, meat and bone meal, red blood cells, lysine hydrochloride, DL-methionine, choice white grease, choline chloride and complete feed. Although PEDV survival varied among feed ingredients and the virus appears to survive the longest in soybean meal, applying a formaldehyde-based liquid treatment can be effective for causing virus inactivation in all ingredients.
Similarly, Trudeau et al. (2017a) evaluated survival of PEDV, TGEV and PDCoV in various feed ingredients and also showed that PEDV virus survived the longest, while TGEV and PDCoV had high survival in soybean meal compared to several other ingredients, including DDGS. These results suggest that soybean meal is a greater risk factor for transmission of coronaviruses via feed than DDGS and other common feed ingredients. Furthermore, thermal treatment at temperatures greater than 70°C inactivated PEDV in complete feed, pre-mix and all ingredients, including DDGS (Trudeau et al., 2017b).
Currently, there is significant concern about the risk of ASFV transmission through imported feed ingredients into the U.S. and other countries (Guinat et al., 2016). If ASFV were to enter the U.S., it has been estimated that economic losses may be about $16.5 billion during the first year of an outbreak (Hayes et al., 2011).
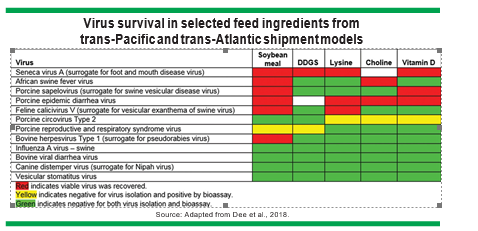
Therefore, Dee et al. (2018) conducted a study to determine the survival of 11 viral pathogens of global significance in feed ingredients under simulated transportation times and environmental conditions across the Pacific Ocean (37 days) and Atlantic Ocean (30 days) to Des Moines, Iowa (Figure). Surrogate viruses with similar characteristics were used to for senecavirus A, porcine sapelovirus, feline calicivirus V, bovine herpes virus Type 1 and canine distemper virus.
Ingredient samples were evaluated using polymerase chain reaction, virus isolation and/or swine bioassays to determine virus survival. Of the five ingredients evaluated, seven viruses survived in soybean meal, while only two - senecavirus A and porcine respiratory and reproductive syndrome virus (PRRSV) — survived in DDGS. Furthermore, four viruses survived in lysine, three survived in choline and four survived in vitamin D. ASFV only survived in soybean meal and choline, but not in DDGS.
These initial results suggest that DDGS is a lower-risk ingredient for transmission of most of the important viruses, and ASFV does not appear to survive in DDGS under the environmental conditions and travel times used in this study.
Conclusion
Our knowledge about the potential health benefits and limitations from feeding DDGS diets to pigs is limited. However, it appears that the relatively high antioxidant compounds and capacity of DDGS may be effective for minimizing oxidative stress in pigs without the need for supplemental vitamin E.Components (beta-glucan, mannans and nucleotides) of yeast cell walls may also provide some gut health and immune system benefits, but no studies have been conducted to directly evaluate their potential contributions.
Furthermore, more studies are needed to improve the understanding of the effects of DDGS on intestinal cell composition, nutrient transporters and receptors, cell differentiation, the immune system, the microbiome and metabolomics. This is critical information to potentially modify diet composition to avoid negative effects from specific pathogen challenges as well as to use DDGS under disease challenge conditions when it may provide some beneficial effects.
Finally, some viruses (PEDV, TGEV, PD- CoV, senecavirus A and PRRSV) appear to survive in DDGS, but the risk of their transmission is much less than in soy- bean meal.
Comments
Post a Comment